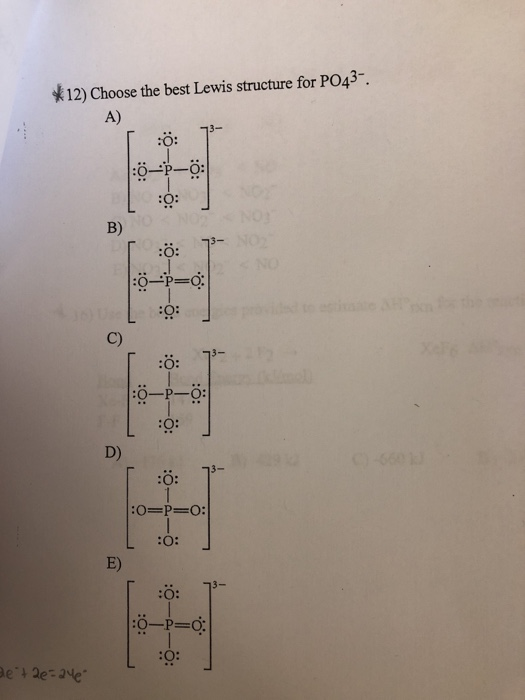
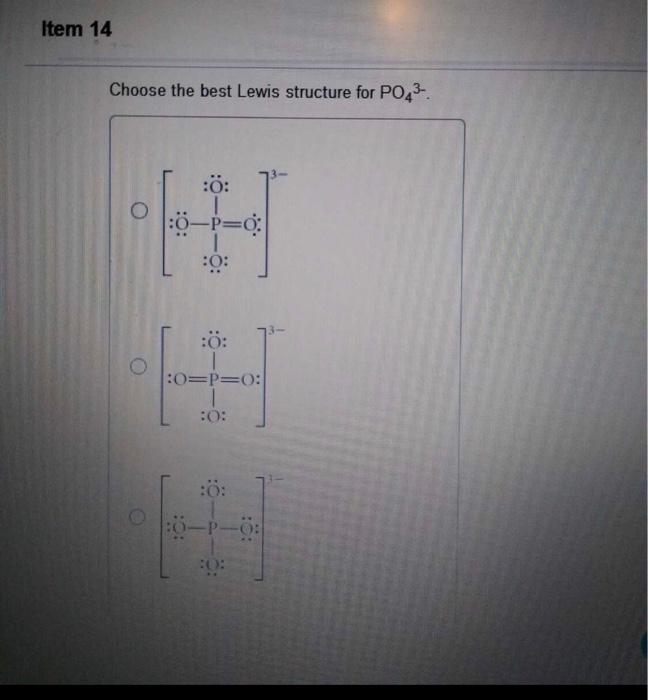
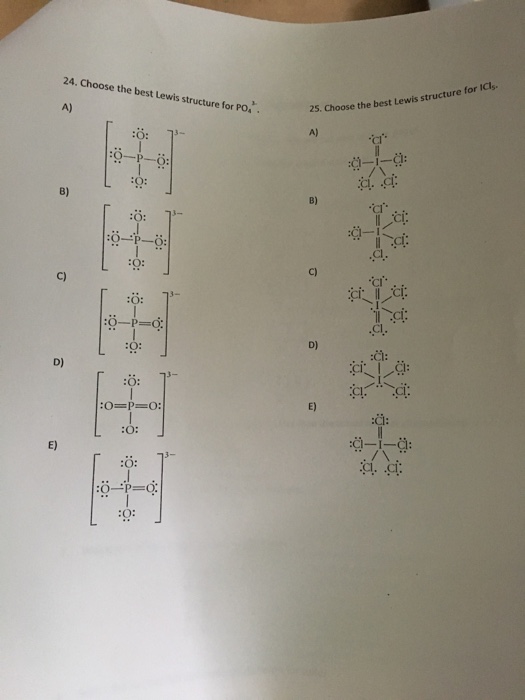
Choose The Best Lewis Structure For Po43
Choose the best lewis structure for po43. For the Lewis structure youll need to have a total charge for the molecule of 3-. Give the hybridization for the O in OF2. This problem has been solved.
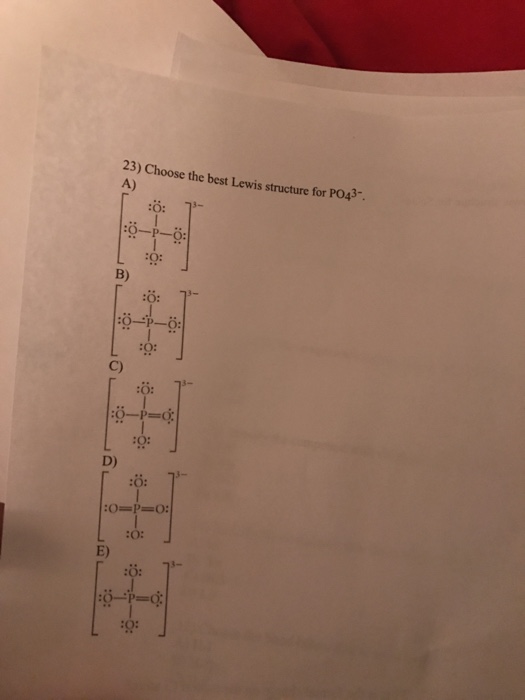
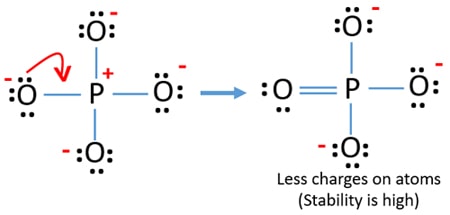
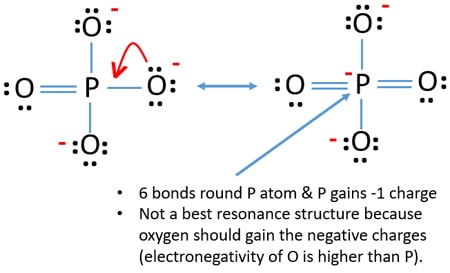
Use information from step 4 and 5 to draw the lewis structure. A all single bonds or B three single bonds and one double bond note this gives rise to four equivalent resonance structures. This is not a good structure because it has five atoms with formal charges.
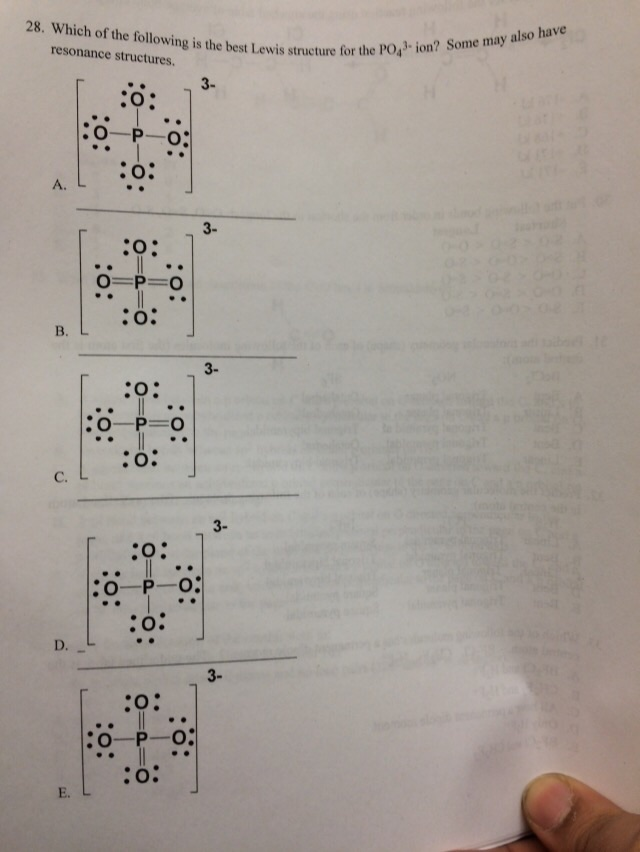
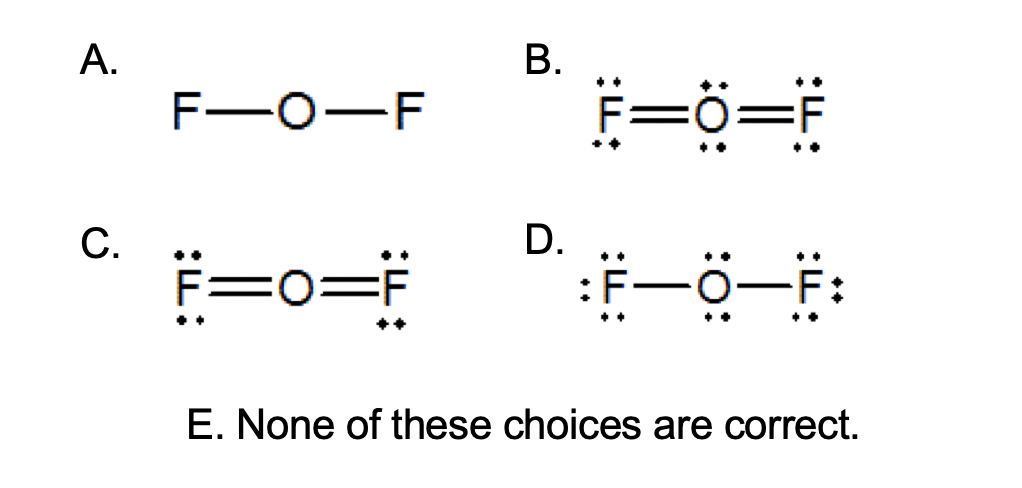
Which of the following statements could chemists use to argue about which is the dominant structure. You can draw five resonance structures for PO_43- but one of them is a minor contributor to the resonance hybrid. Give the electron geometry molecular geometry and hybridization for XeF4.
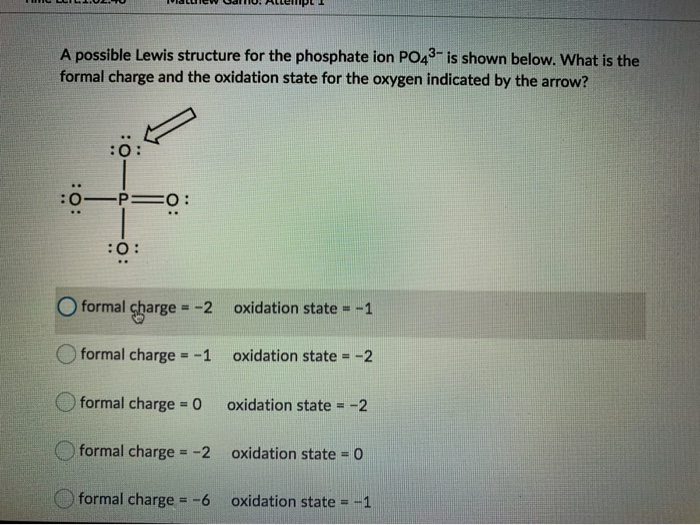
Draw Lewis structures and assign formal charges for structures with. Type your question here Choose the best Lewis structure forPO43-. Choose the best Lewis structure for XeI2.
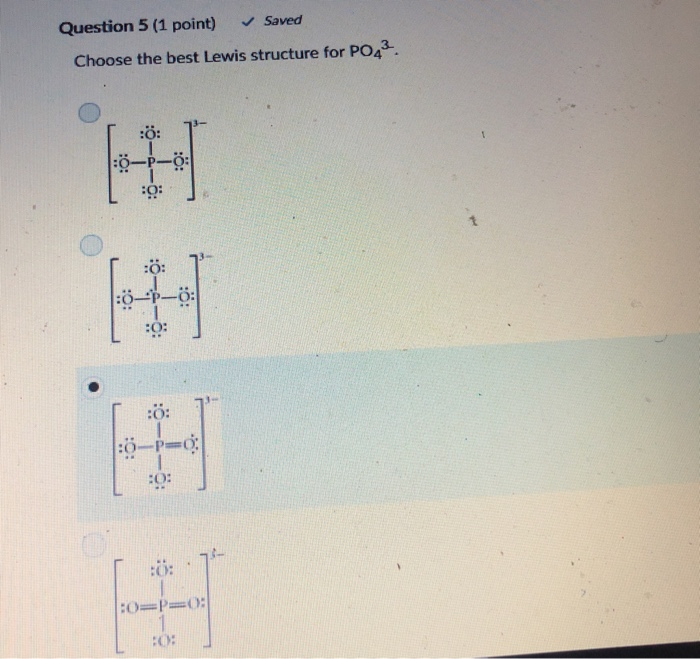
Place a P atom in the center. It also discusses the formal charge and resonance struc. Choose the best Lewis structure for PO43-.
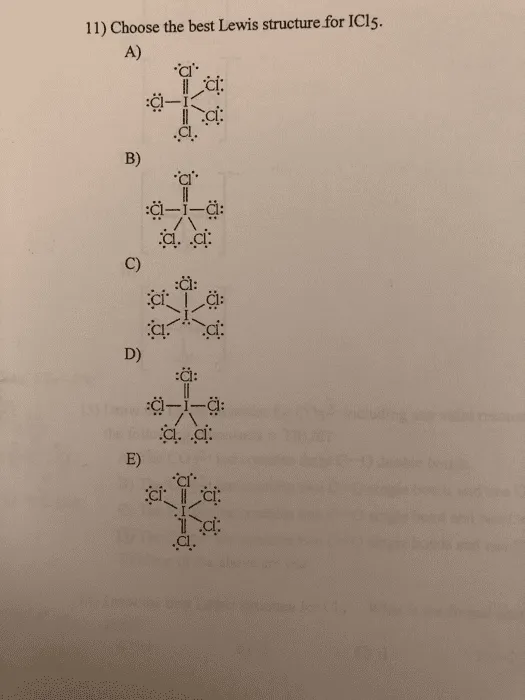
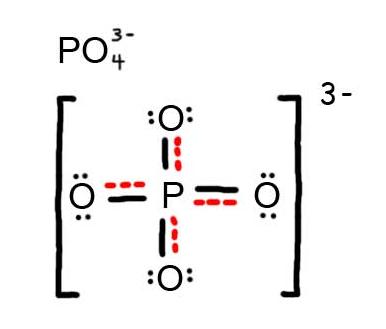
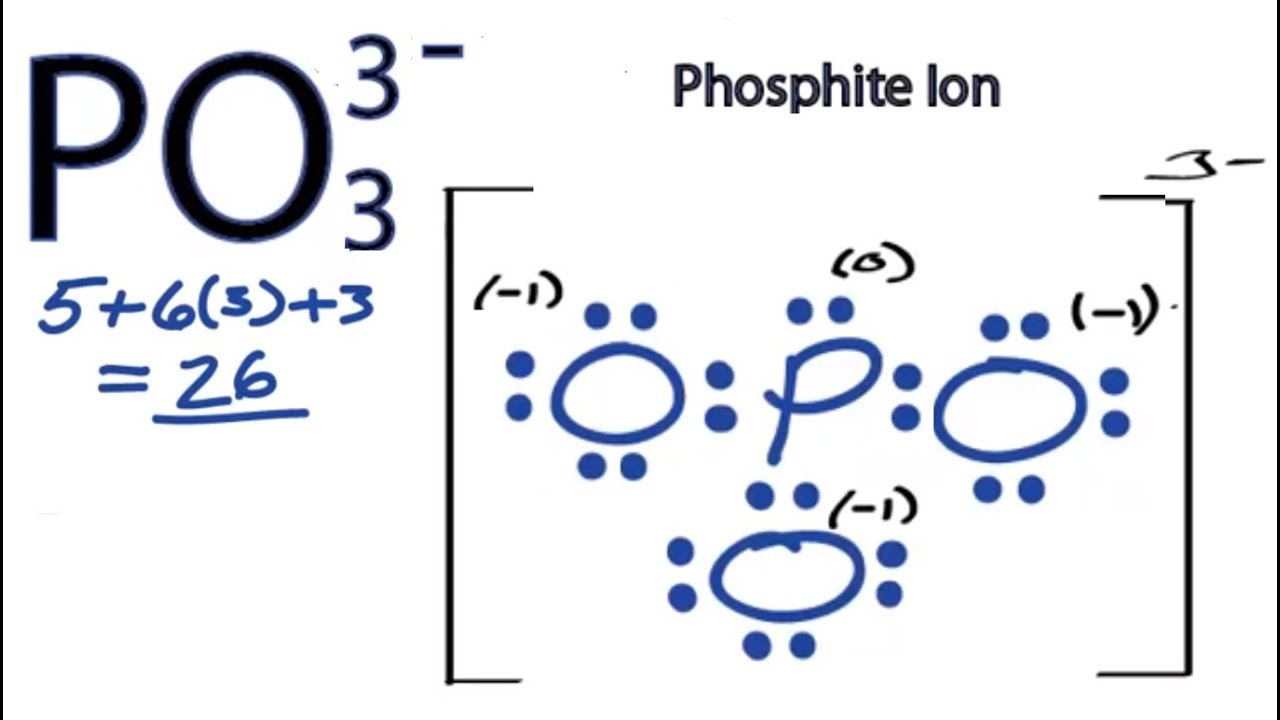
Choose the best Lewis structure for SF4. SF6 XeF4 and PO4 3. Lewis structure of PO 43- ion In the lewis structure of PO 43- three is a double bond between phosphorous atom and one oxygen atom.
Single bond this P atom to 3 separate O atoms. Expert Answer 100 11 ratings Previous question Next question Transcribed Image Text from this Question.
Watch your formal charges youll get the correct structure.
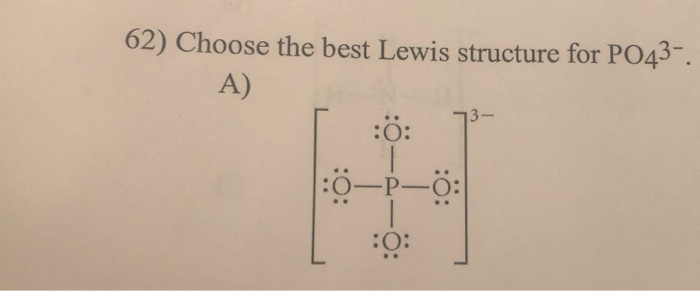
For the Lewis structure youll need to have a total charge for the molecule of 3-. Get the detailed answer. Lewis dot structure of PO 4 3-Alternatively a dot method can be used to draw the lewis structure. SF6 XeF4 and PO4 3. Give the number of valence electrons for SO42-32. Between other oxygen atoms there are only single bonds with phosphorous atom. Thats the Lewis structure for PO4 3-. Watch your formal charges youll get the correct structure. It also discusses the formal charge and resonance struc.
You can draw five resonance structures for PO_43- but one of them is a minor contributor to the resonance hybrid. Give the hybridization for the O in OF2. Draw Lewis structures and assign formal charges for structures with. Thats the Lewis structure for PO4 3-. Between other oxygen atoms there are only single bonds with phosphorous atom. Consider the phosphate ion PO43-. Which of the following statements could chemists use to argue about which is the dominant structure.





























Post a Comment for "Choose The Best Lewis Structure For Po43"